As a result, they are employed as normal states or reference points for calculating the various thermodynamic characteristics of these components. The standard states of atomic elements are stated in relation to the most stable and sober allotrope for every element for example, white tin and graphite are some of the most stable and uniformed allotropes of tin and carbon, respectively. H o rxn = n H o f (products) – m H f o (reactants) However, most tables of thermodynamic values are constructed at certain temperatures, most typically 298.15 K (exactly 25☌) or, slightly less frequently, 273.15 K (exactly 0☌). The standard gas state is traditionally selected to be 1 bar for an ideal gas, independent of temperature. Solely, the temperature cannot be a part of a standard state definition. A pressure of 1 bar (101.3 kilopascals) is established as the norm. 
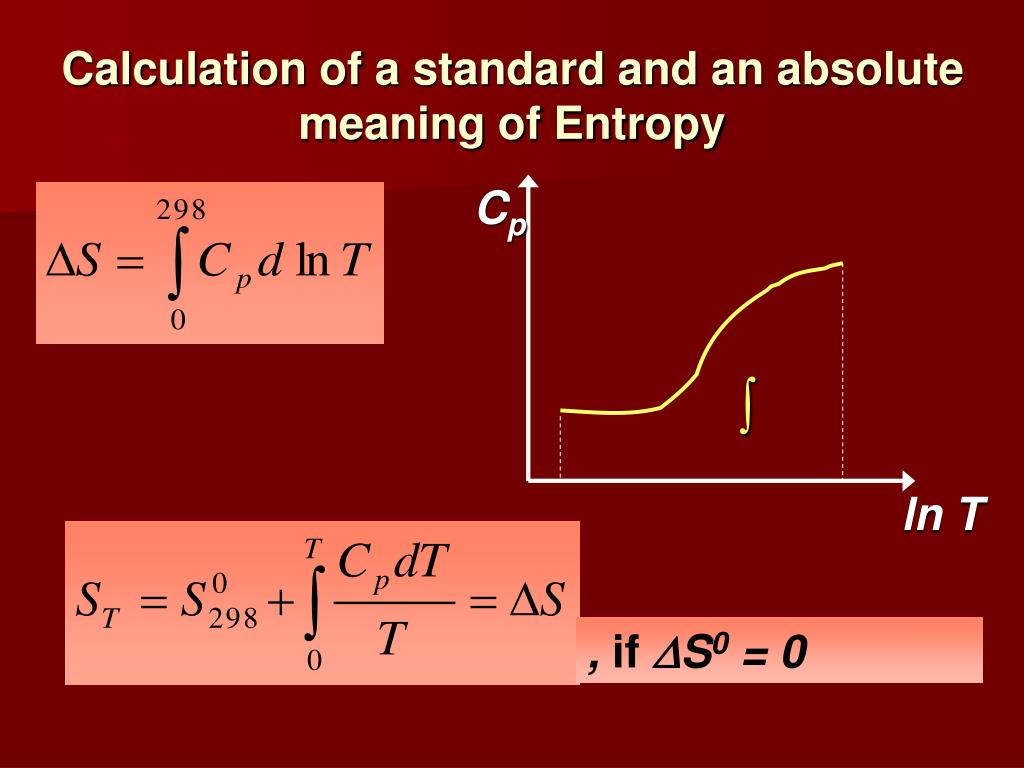
Although the International Union of Pure and Applied Chemistry (IUPAC) suggests a typically decided standard-level for widespread usage, the choice of the standard state is arbitrary in theory. In chemistry, a material’s standard state, whether a pure substance or combination, is a reference point or a solution utilised to understand and determine its characteristics under varied situations. By adding the known temperatures of formation or combustion for the process stages, one may determine the heat absorbed or evolved in any chemical reaction using Hess’s equation of heat summation. S° f for chemical compounds may be computed using the S° values of the elements from which the compound is created.Īn element’s creation heat is arbitrarily assigned a value of zero. The entropy formation of one mole of a compound under standard circumstances (S° f ) is defined as the entropy formation of one mole of a compound under standard conditions. The standard entropy change of a chemical process or reaction is stated when S° of several substances are known. Where ΔS° is the standard entropy change of the reaction. At any temperature over 0°K, the conventional entropies of all substances, whether elements or compounds, are always positive. The standard entropy (S°) is the absolute entropy of a pure material at 25☌ (298 K) and 1 atm pressure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
